Solved) - NRT The Ideal Gas Equation States That Pi Where P Is The Pressure, (1 Answer)
amp;#160;NRT The Ideal Gas Equation States That Pi Where P Is The Pressure, N Is The V Number Of Moles Of Gas, R= .08206, T Is The Temperature (In Degrees Kelvin), And V Is The Volume Of The Gas. At High Pressure, A More Accurate Equation Is The Van NRT

Derivation of The Chemical Potential of a Compound - Handout

Osmosis and Osmotic Pressure: Definition, Formulas, Proof, Videos, Q&A

PPT - Ideal Gas Law PowerPoint Presentation, free download - ID
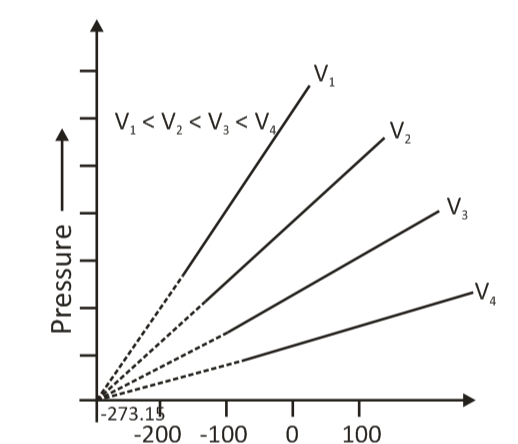
Gay Lussac's Law of Thermodynamics

Van der Waals Equation, Definition & Examples - Lesson

Maharashtra Board Class 11 Chemistry Solutions Chapter 10 States

What is the Ideal Gas Law?

Vander Waals Equation

1st PUC Chemistry Question Bank Chapter 5 States of Matter - KSEEB Solutions
What is the ideal gas law? - Quora
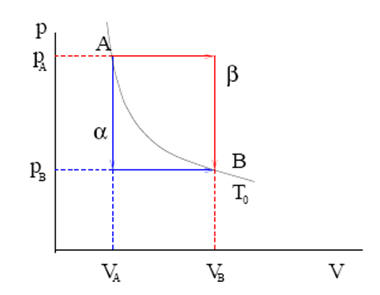
1st law

Gas Laws - Equations and Formulas

Compressibility factor, Z of a gas is given as Z= frac { pV }{ nRT

Equation of State, Glenn Research Center