Medical device regulations, classification & submissions
$ 11.99
In stock
4.6
(391)

Medical device regulations vary in Canada, the U.S. & the EU. Risk-based classification systems determine data requirements for regulatory oversight for medical devices. MaRS
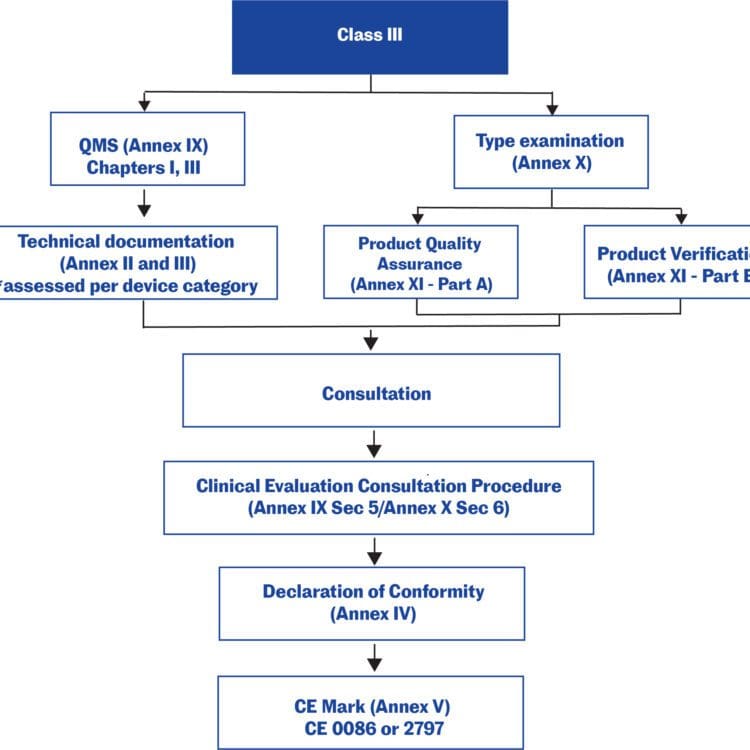
Medical device regulations, classification & submissions

Medical device regulations, classification & submissions
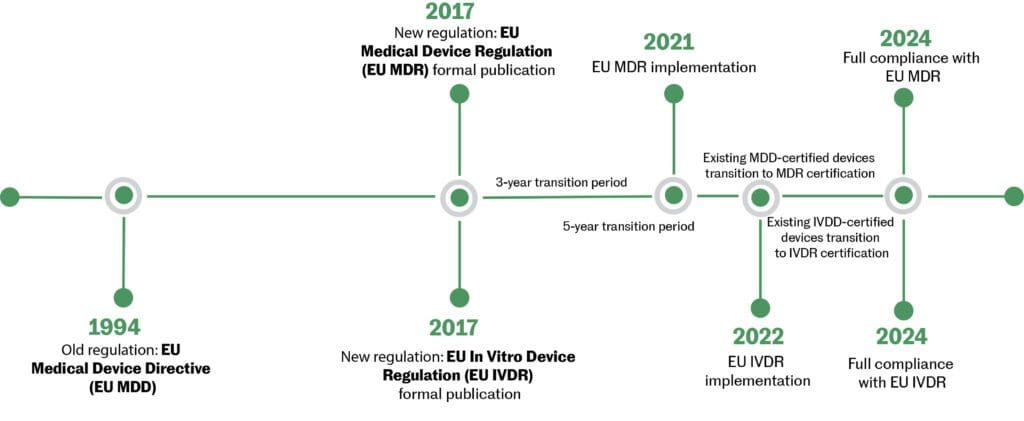
Medical device regulations, classification & submissions

Medical device regulations, classification & submissions
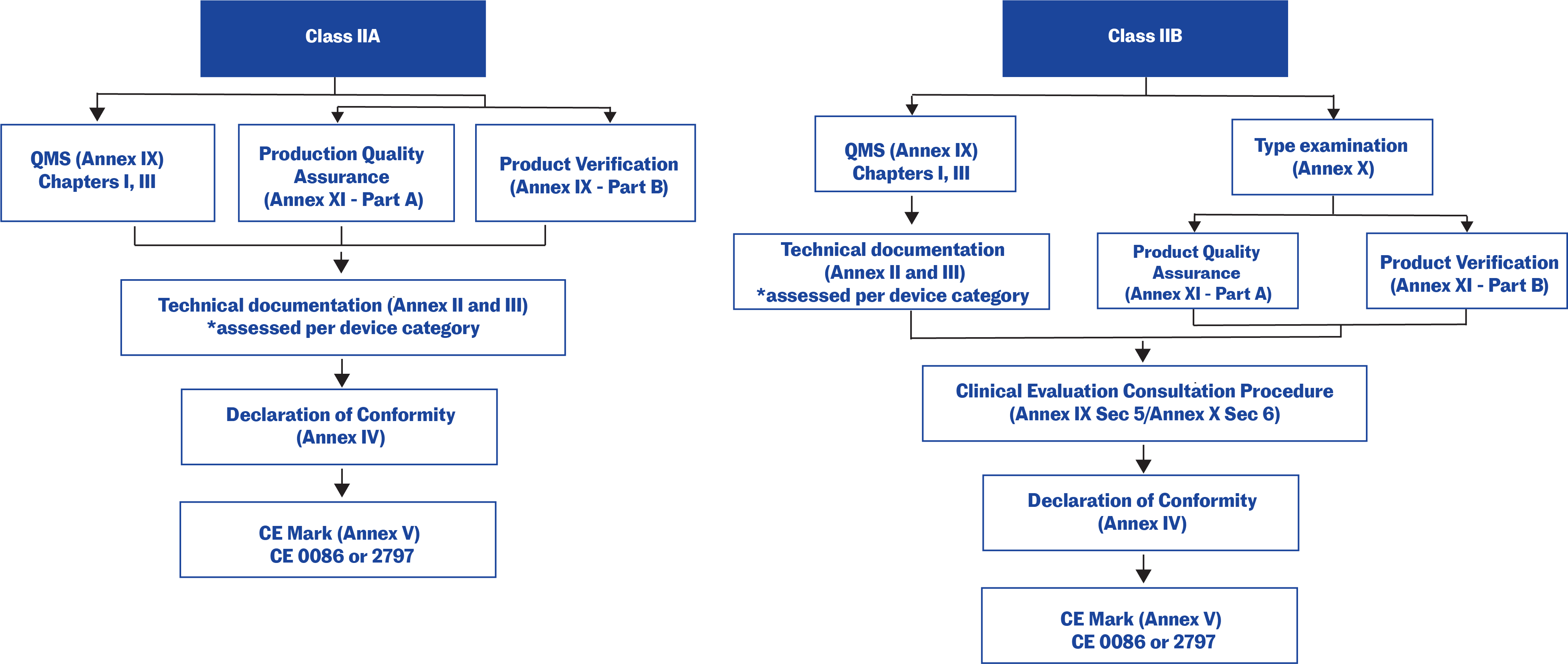
Medical device regulations, classification & submissions