High-Power, Short-Duration Ablation in the Treatment of Atrial

Catheter ablation is the cornerstone of the rhythm control treatment of atrial fibrillation (AF). During this procedure, creating a contiguous and durable lesion set is essential to achieve good long-term results. Radiofrequency lesions are created in two phases: resistive and conductive heating. The ablation catheters and the generators have undergone impressive technical developments to enable homogenous and good-quality lesion creation. Despite recent years’ achievements, the durable isolation of the pulmonary veins remains a challenge. These days, intensive research aims to evaluate the role of high-power radiofrequency applications in the treatment of patients with cardiac arrhythmias. The use of high-power, short-duration applications might result in a uniform, transmural lesion set. It is associated with shorter procedure time, shorter left atrial, and fluoroscopy time than low-power ablation. This technique was also associated with a better clinical outcome, possibly due to the better durability of lesions. Multiple clinical studies have proven the safety and efficacy of high-power, short-duration PVI.

High-Power Short Duration Ablation for Pulmonary Vein Isolation

PDF) The superiority of high‐power short‐duration radiofrequency

PDF) The superiority of high‐power short‐duration radiofrequency

Pulsed‐field ablation combined with ultrahigh‐density mapping in
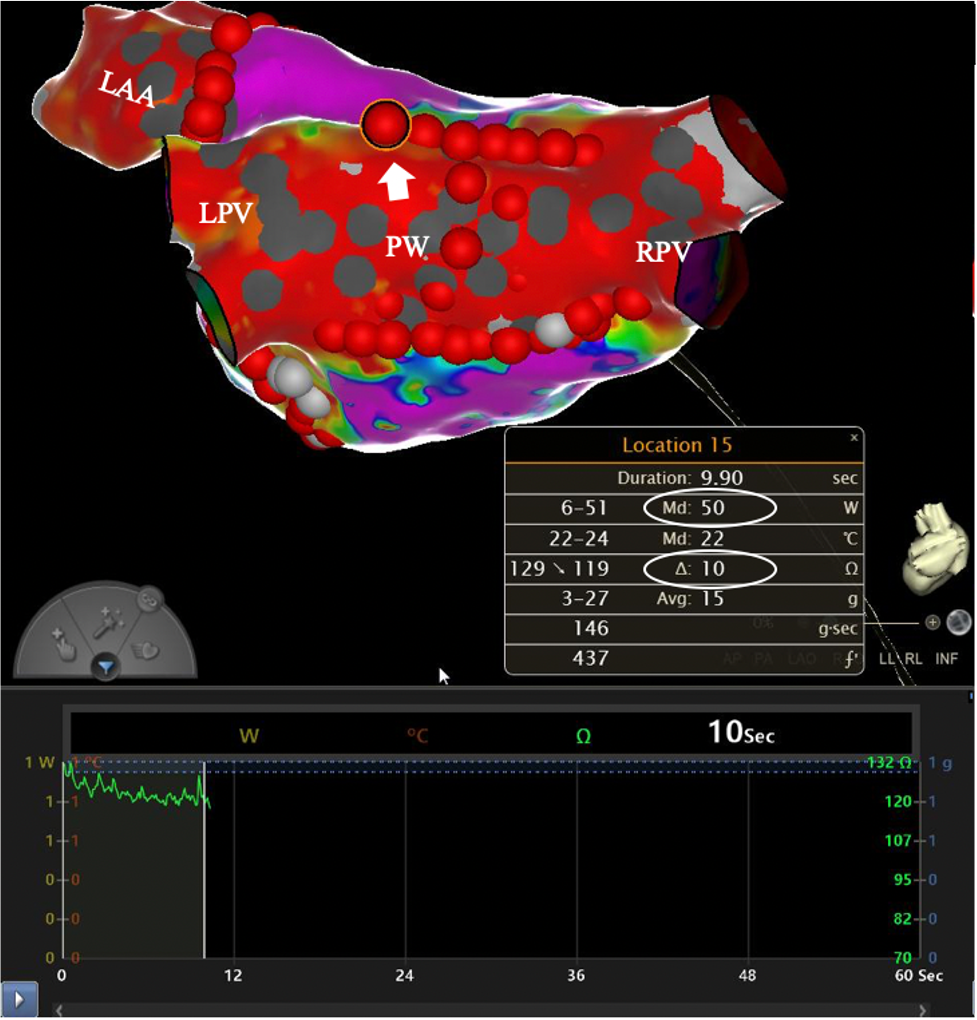
High-Power Short-Duration Ablation of Paroxysmal and Persistent

High-power short-duration versus standard-power standard-duration

Network Meta-Analysis and Systematic Review Comparing Efficacy and

Pulmonary vein isolation for atrial fibrillation using true high

Very high-power short-duration temperature-controlled ablation

PDF) Left Atrial Posterior Wall Isolation with Pulsed Field

PDF) The role of local impedance drop in the acute lesion efficacy

Rhythm Control of Atrial Fibrillation: The Earlier the Better
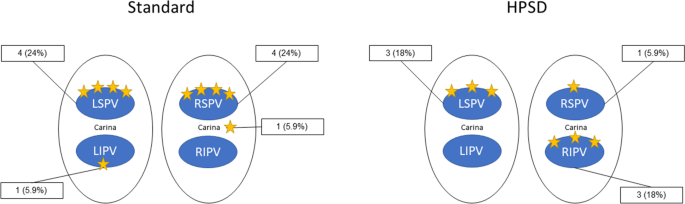
High-power short-duration ablation index–guided pulmonary vein

Reproducibility of the CLOSE protocol (the VISTAX trial) [8