Suggested Book Problems Even answers - CHEMISTRY A Molecular Approach, Nivaldo Tro, Fourth Edition - Studocu

Share free summaries, lecture notes, exam prep and more!!
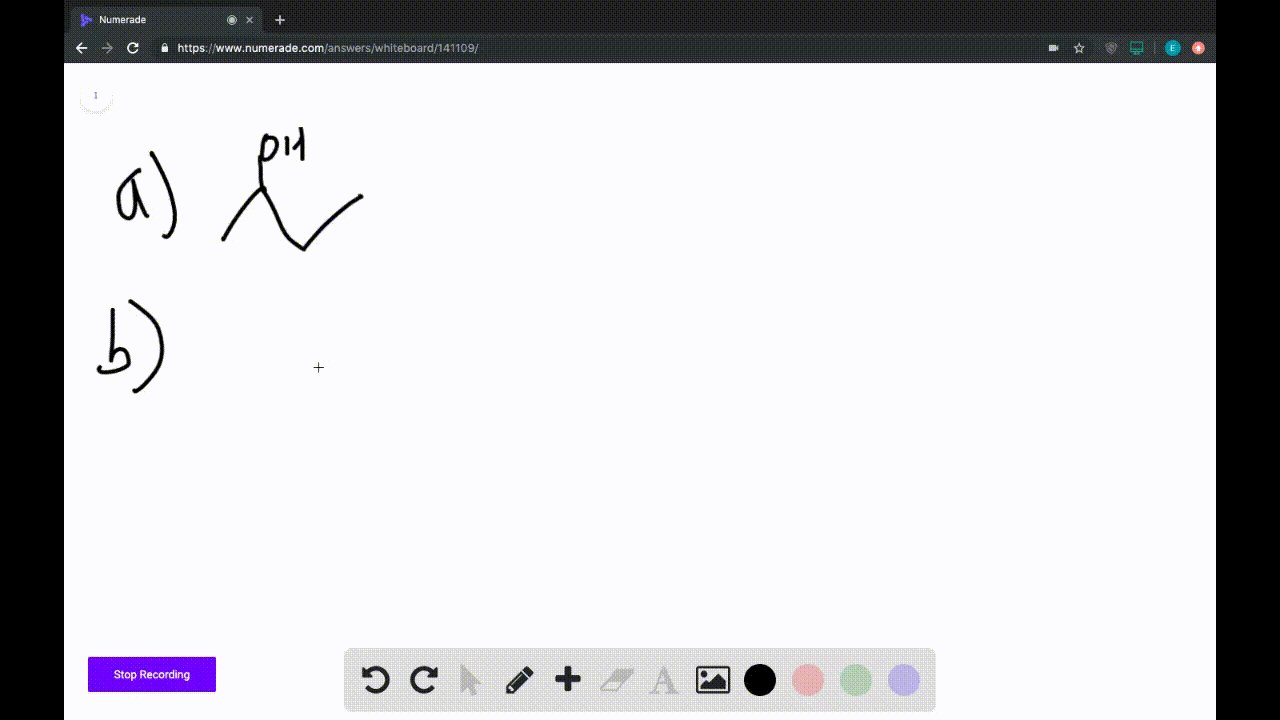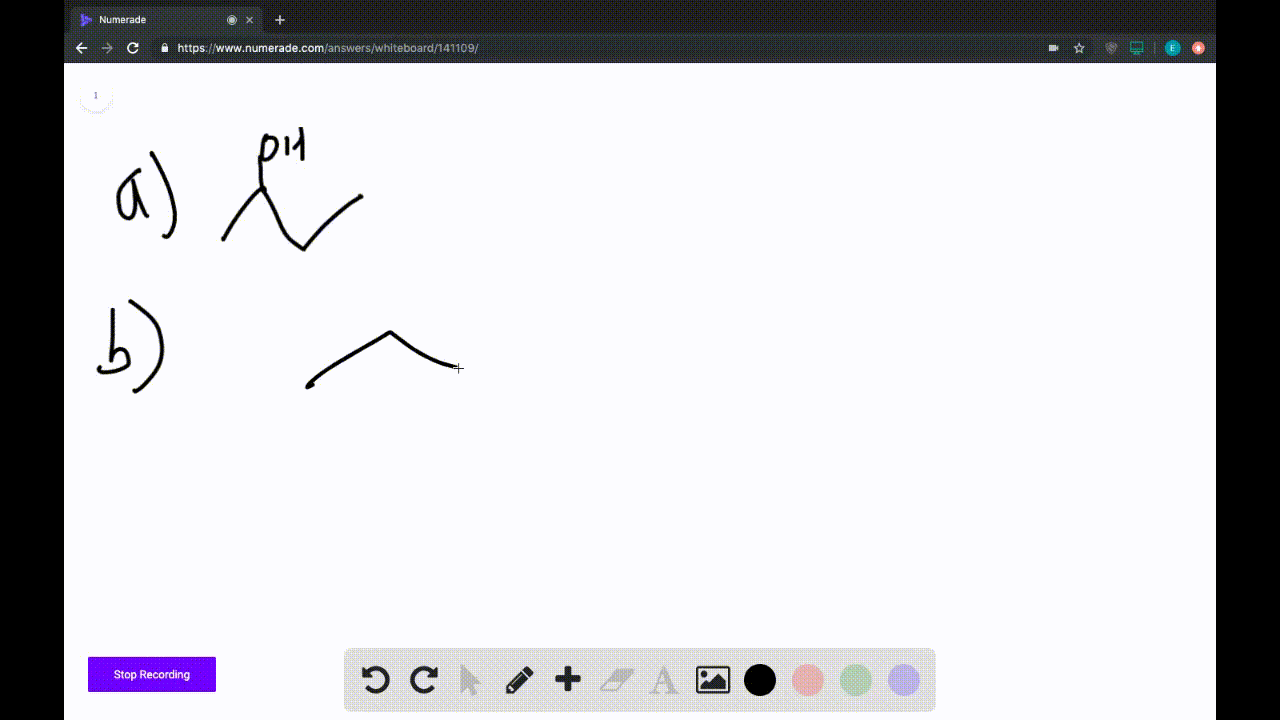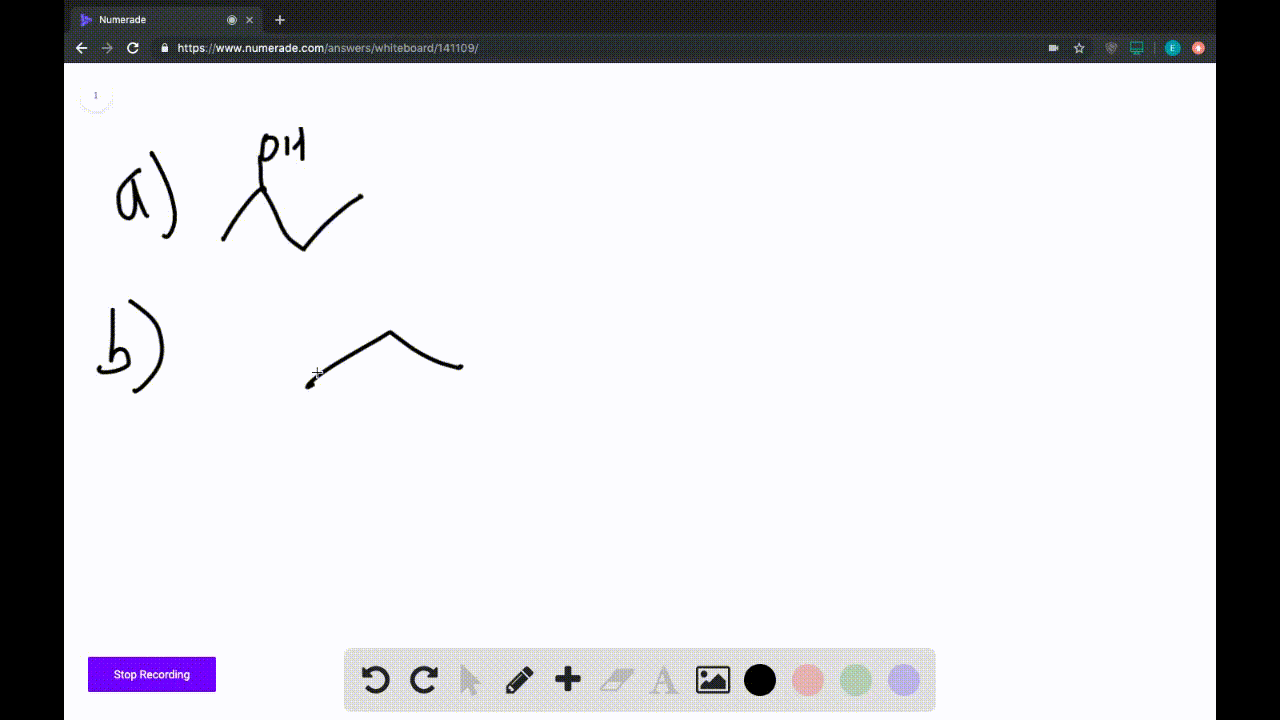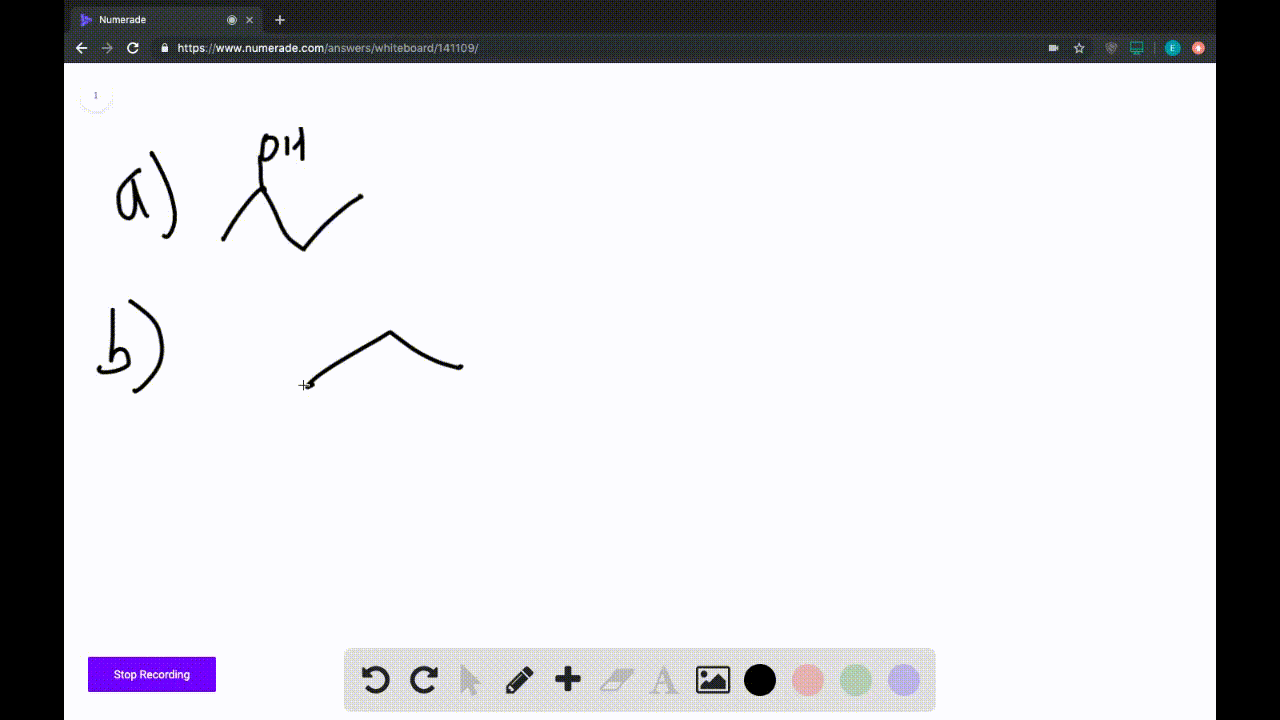
A) Avogadro's Law.
B) Ideal Gas Law.
C) Charles's Law.
D) Boyle's Law.
E) Dalton's Law.
A) Vapor pressure increases with temperature.
B) Hydrogen bonds are stronger than covalent bonds.
C) Intermolecular forces hold the atoms in molecules together.
D) Dispersion forces are generally stronger than dipole-dipole forces.
E) None of the above is true.
A) hydrogen bonding.
B) ion-dipole forces.
C) dipole-dipole forces.
D) dispersion forces.
E) ionic forces.
Highlight, take notes, and search in the book In this edition, page numbers are just like the physical edition

Chemistry: A Molecular Approach
Solutions for Chemistry A Molecular Approach 4th by Nivaldo J. Tro, Book solutions

Chemistry: A Molecular Approach: Tro, Nivaldo: 9780134874371: : Books
Solutions for Chemistry A Molecular Approach 4th by Nivaldo J. Tro, Book solutions

Chemistry A Molecular Approach 4th Edition Ebook PDF

Chemistry: A Molecular Approach (4th Edition) Textbook Solutions

A01 TRO4904 03 SSM FM - Extra - Copyright © 2020 Pearson Canada Inc. SELECTED SOLUTIONS MANUAL Noel - Studocu

Chemistry: A Molecular Approach (4th Edition) Textbook Solutions

Grade 12 Q2 General Chemistry 2 LAS, PDF, Entropy

Syllabus CHMA11 Winter 2022 Final winter semester - Introductory Chemistry II — CHMA11 Winter 2022 - Studocu

Chemistry: A Molecular Approach (4th Edition) Textbook Solutions

Chemistry A Molecular Approach 4th Edition Ebook PDF

Principles of Chemistry 3e by Nivaldo J. Tro - 3 Li6 11 Na 22 19 K 39 37 Rb 85 55 Cs 132. [223] 87 - Studocu

Solutions for Chemistry A Molecular Approach 4th by Nivaldo J. Tro, Book solutions