What is the mass of glucose required to produce 44g of C{O_{2'}} on complete combustion?30g45g60g22g

Click here:point_up_2:to get an answer to your question :writing_hand:what is the mass of glucose required to produce 44g of co2 on complete
Click here👆to get an answer to your question ✍️ What is the mass of glucose required to produce 44g of C-O-2- on complete combustion-30g45g60g22g

Solved Question 2 (3 points) Saved The combustion of sugar

What mass of glucose is required to produce 88 g of CO2 on complete combustion?

Solved -. The atomic mass of oxygen (O2) is 16.00 g/mol.

Solved A 4.50 g sample of sugar C5H1005 (molar mass= 150.0
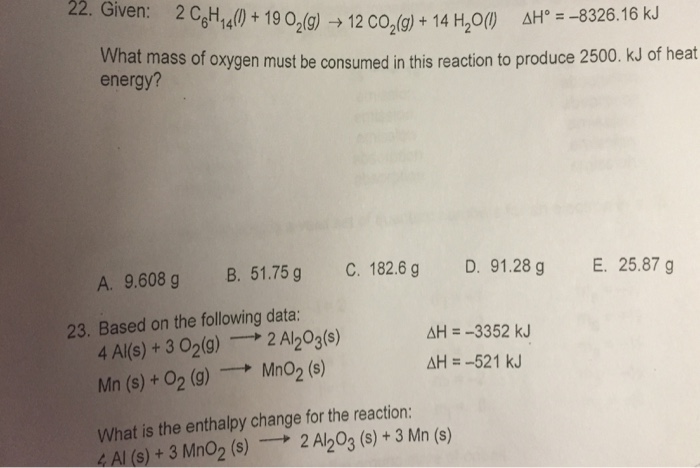
Solved 22 Given: 2 C, H140+19 C2 2 G c6H14() + 1902(g) → 12


What is the mass of glucose required to produce 44 g of CO_(2), on complete combustion?, 12

SOLVED: For the combustion reaction: C6H12O6 + 6 O2 -> 6 H2O + 6 CO2 If you burn 100 g of glucose (C6H12O6), how many grams of CO2 are produced? Problem-solving pathway

What is the mass of glucose required to produce 44 g of CO_(2), on complete combustion?, 12
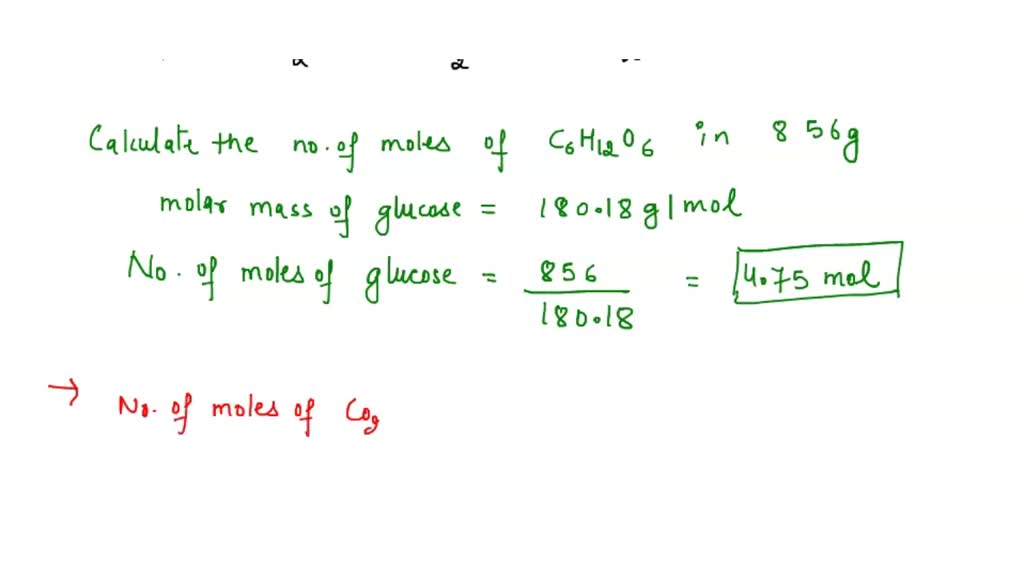
SOLVED: If during a certain period a person consumes 856 g of glucose, what is the mass of CO2 produced? Perform the relevant calculations and attach them to this question. C6H12O6 +