The compression factor (Z) Co, 7°C and 100 atm is 0.21. Calculate

Click here:point_up_2:to get an answer to your question :writing_hand:the compression factor z for co at 7c and 100atm is 021 calculate the volume
Click here👆to get an answer to your question ✍️ The compression factor -Z- Co- 7-C and 100 atm is 0-21- Calculate the volume of a 4 mole sample of co- same temperature and pressure -use R - 0-08 L- atm-K-mol -1- 0-192 -2- 0-05 L -3- 0-38 L -4- 0-44 L closed container can be

Compressibility Factor Z
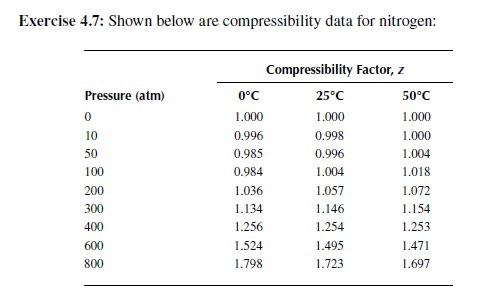
Solved Exercise 4.7: Shown below are compressibility data

Chemical Thermodynamics
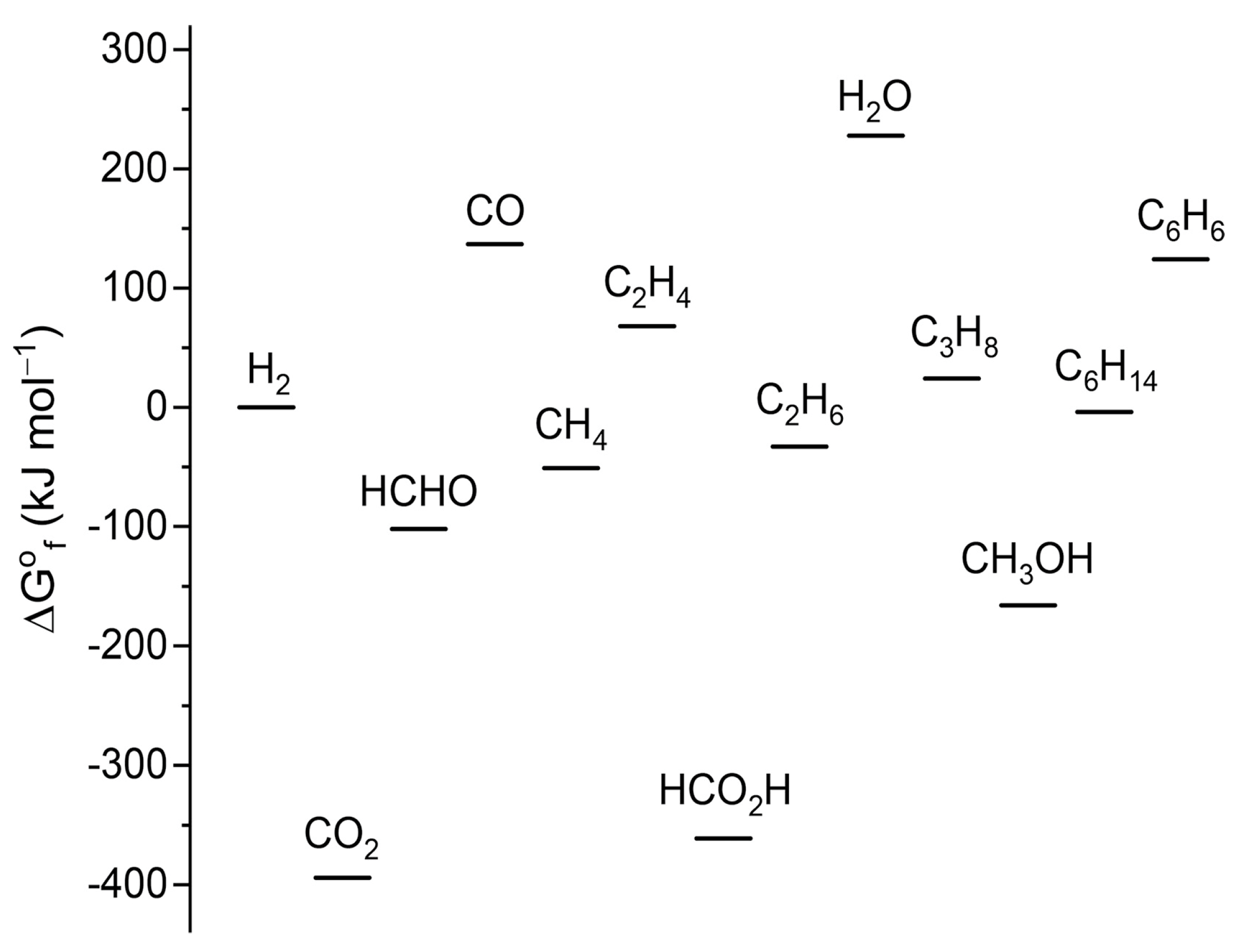
Inorganics, Free Full-Text

Sheet - 01 - Real Gas, PDF, Gases

CH 6. State of Matter (Chem +1) PDF, PDF, Gases
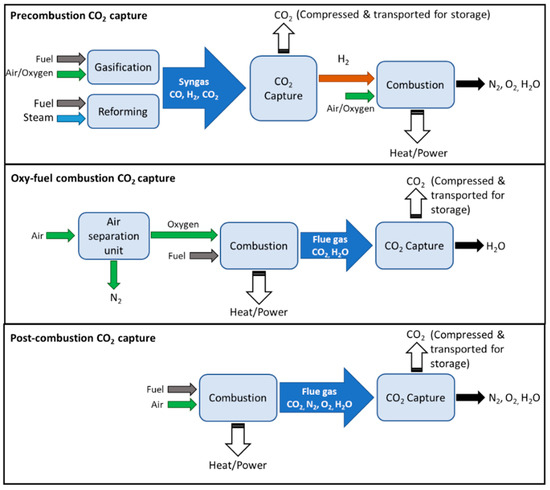
Integration of CO2 Capture and Conversion by Employing Metal
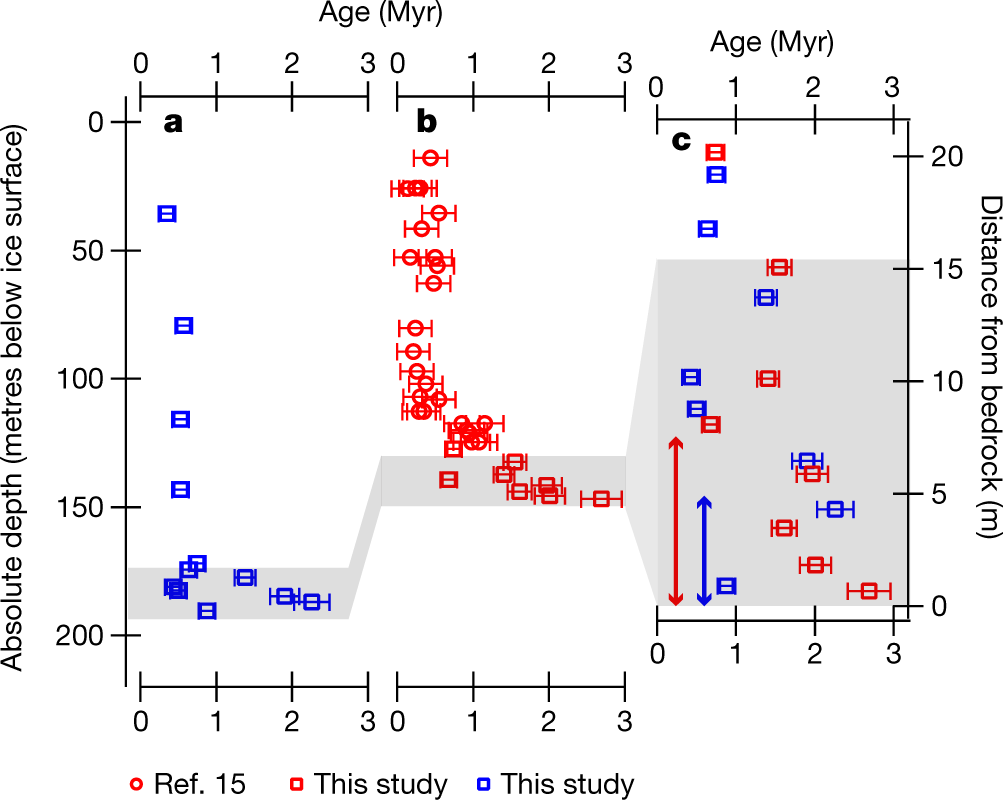
Two-million-year-old snapshots of atmospheric gases from Antarctic

Peter Atkins Julio de Paula Ron Friedman Physical Chemistry Quanta

Bansal classes chemistry study material for iit jee by S.Dharmaraj
Processes, Free Full-Text

McMurry and Fay On-Line Chapters

Gas dynamics and jet propulsion – presentationof problemsanswers