Preparation of functionalized nanocomposites Fe3O4@SiO2-3-aminopropyltrimethoxysilane and its adsorption to Pb(Ⅱ)
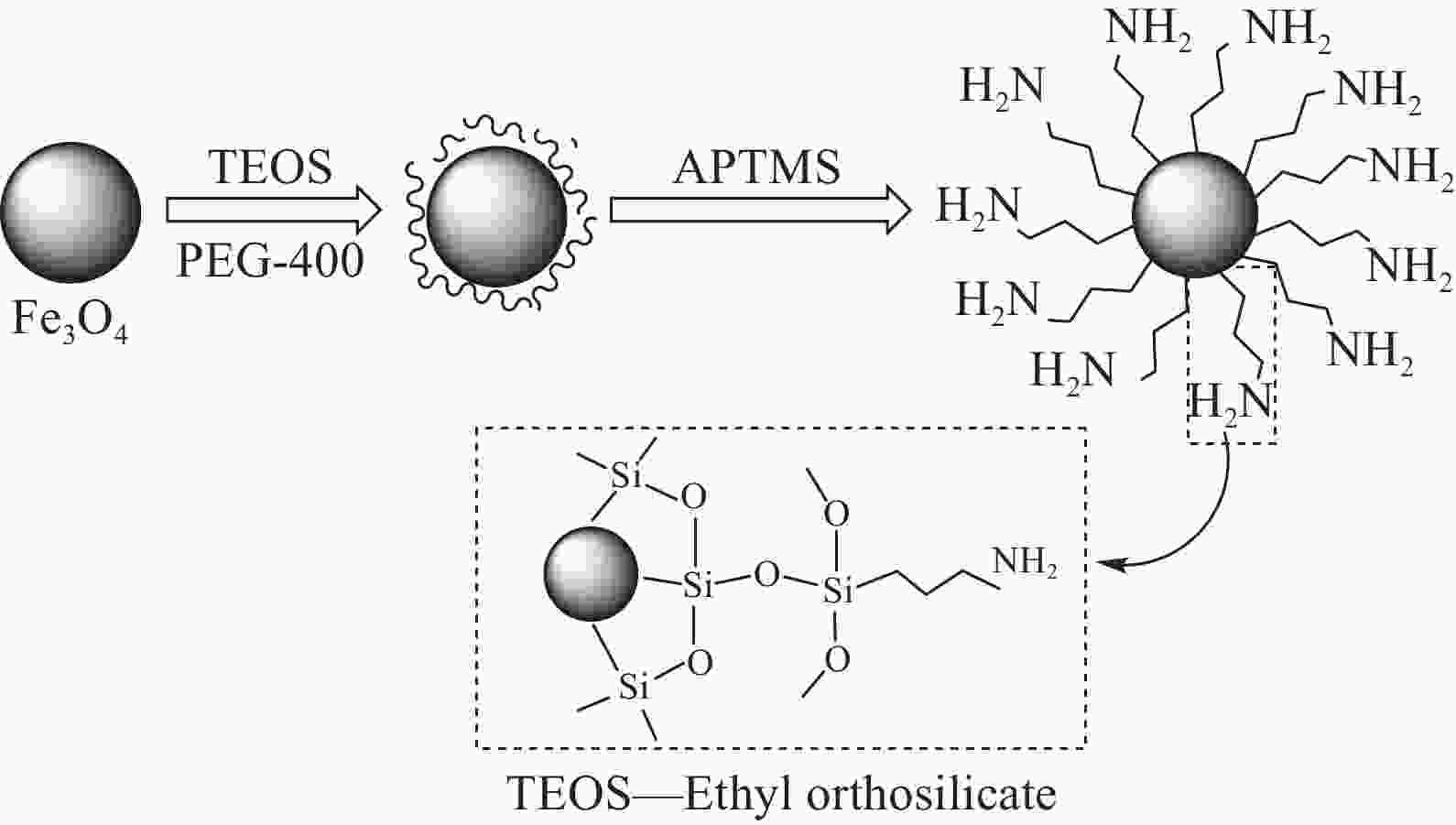
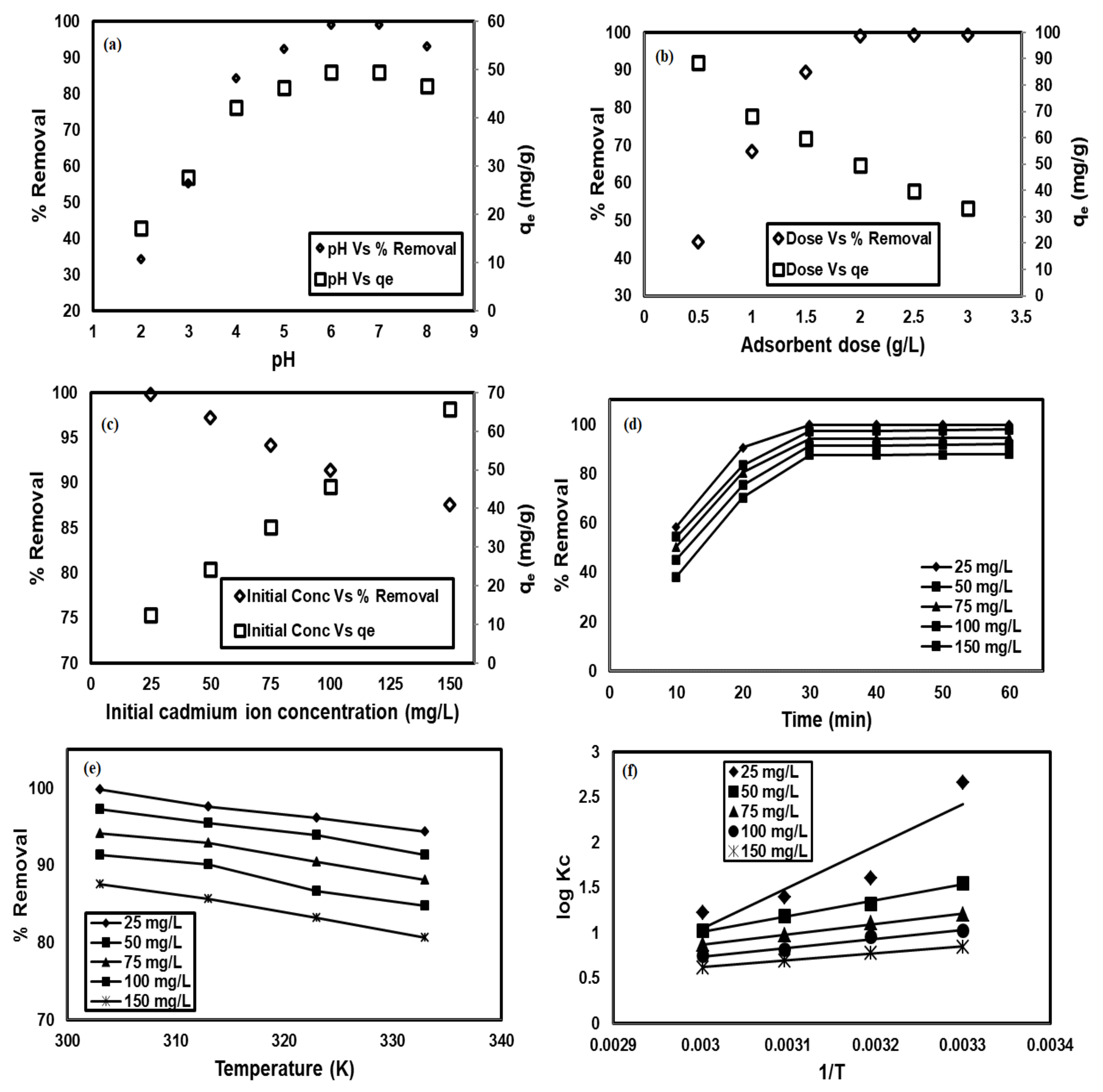
In order to solve the indefects that magnetic nano-Fe<sub>3</sub>O<sub>4</sub> particles were corroded and agglomerated easily, functional modification was carried out. FeCl<sub>3</sub> and FeSO<sub>4</sub> were used as raw materials and ammonia as preci-pitant in the presence of ultrasonic irradiation, then functionalized by ethyl orthosilicate (TEOS) and 3-aminopropyltrimethoxysilane (APTMS) to prepare SiO<sub>2</sub>-coated amino-functional nanocomposites Fe<sub>3</sub>O<sub>4</sub>@SiO<sub>2</sub>-APTMS. The magnetic nanocomposites were characterized by TEM, FTIR, VSM, TGA, low temperature nitrogen adsorption and XRD, etc. The characterized results show that the magnetic nanocomposites prepared by ultrasonic strengthening method have the characteristics of strong magnetic response, strong acid and alkali resistance, high dispersion, large specific surface area and small particle size.Meanwhile, the adsorption effects of magnetic nanocomposites for Pb(Ⅱ) were investigated. The results show that the initial pH value of the solution and the dosage of adsorbent have greatest effects on the adsorption effect of Pb(Ⅱ) with the initial pH value of the solution 5.86 and the dosage of adsorbent 1.0-1.5 g·L<sup>−1</sup>. The Langmuir model is suitable for simulating the isothermal adsorption process, and the adsorption process is a spontaneous process when Gibbs free energy change <i>∆G</i><sup>0</sup><0. The adsorption behavior of Pb(Ⅱ) can be well described by quasi-second-order kinetics on the composites,<i> </i>Quasi-second-order kinetic constant<i> k</i><sub>2</sub>=0.0401 g·mg<sup>−1</sup>·min<sup>−1</sup>,<i> </i>equilibrium adsorption capacity<i> q</i><sub>e</sub>=80.041 mg·g<sup>−1</sup>; it is speculated that the adsorption mechanism is mainly complex adsorption and ion exchange.
Sustainability, Free Full-Text

Poly (amidoamine) generation 6 functionalized Fe3O4@SiO2/GPTMS core–shell magnetic NPs as a new adsorbent for Arsenite adsorption: kinetic, isotherm and thermodynamic studies

Multifunctional nanocomposites Fe3O4@SiO2-EDTA for Pb(II) and Cu(II) removal from aqueous solutions - ScienceDirect

Polymers, Free Full-Text
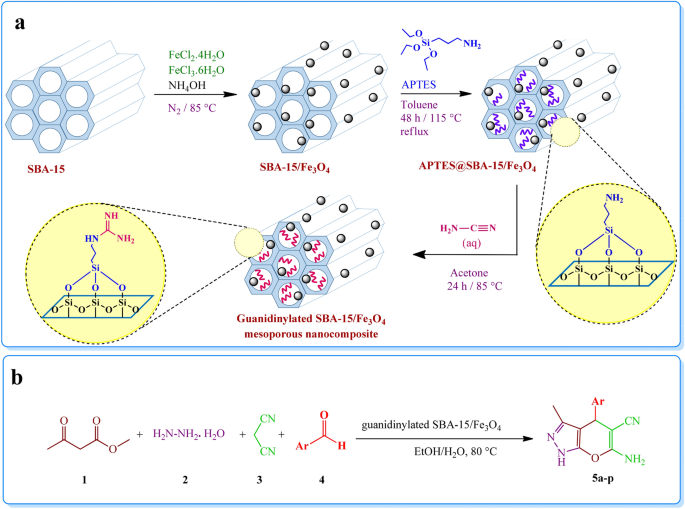
Guanidinylated SBA-15/Fe3O4 mesoporous nanocomposite as an efficient catalyst for the synthesis of pyranopyrazole derivatives

Functionalized magnetic nanoparticles Fe3O4@SiO2@PTA (PTA = (2-pyrimidylthio)acetic acid) for efficient removal of mercury from water - ScienceDirect

Simultaneous fluorescence response and adsorption of functionalized Fe3O4@ SiO2 nanoparticles to Cd2+, Zn2+ and Cu2+ - ScienceDirect

Figure 2 from Facile preparation of magnetic mesoporous MnFe2O4@SiO2-CTAB composites for Cr(VI) adsorption and reduction.
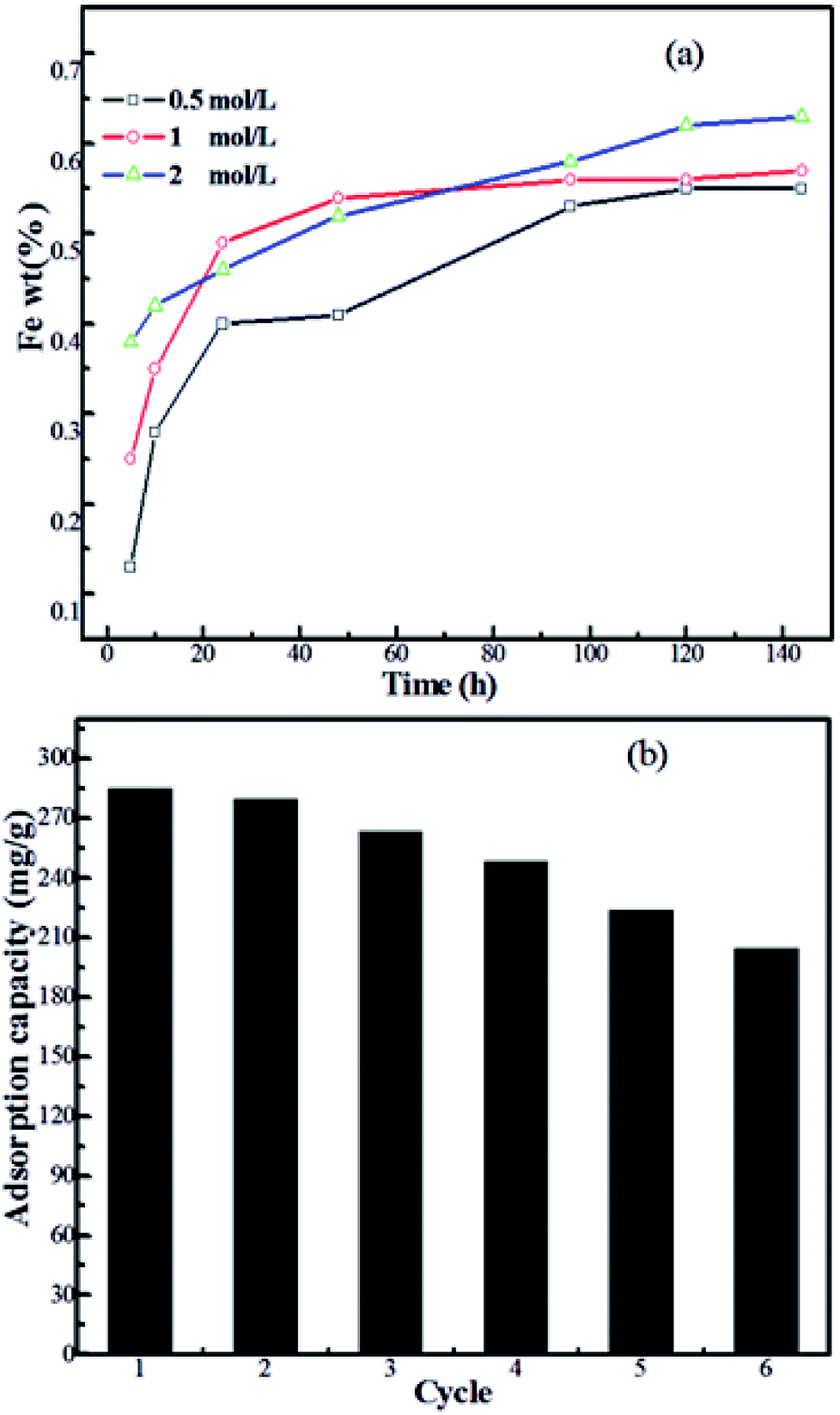
Novel magnetically separable anhydride-functionalized Fe 3 O 4 @SiO 2 @PEI-NTDA nanoparticles as effective adsorbents: synthesis, stability and recycl - RSC Advances (RSC Publishing) DOI:10.1039/C8RA10310K

Adsorption of Cr(Ⅵ) polluted water by Fe3O4@SiO2-APTMS nanocomposites prepared in the presence of ultrasonic irradiation for sustainable water resources utilization - ScienceDirect

Triethylenetetramine-modified hollow Fe3O4/SiO2/chitosan magnetic nanocomposites for removal of Cr(VI) ions with high adsorption capacity and rapid rate - ScienceDirect

PDF] Facile preparation of magnetic mesoporous MnFe2O4@SiO2-CTAB composites for Cr(VI) adsorption and reduction.

Sustainability, Free Full-Text