Health-related quality of life and quality-adjusted progression
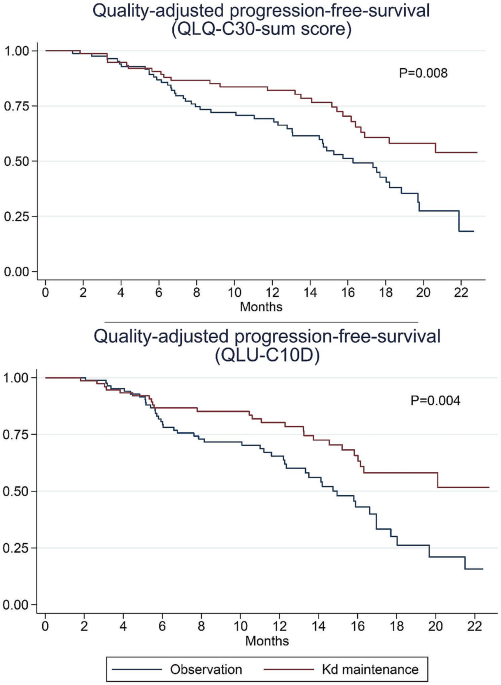
Background Decisions regarding maintenance therapy in patients with multiple myeloma should be based on both treatment efficacy and health-related quality of life (HRQL) consequences. In the CARFI trial, patients with first relapse of multiple myeloma underwent salvage autologous stem cell transplantation (salvage ASCT) before randomization to carfilzomib-dexamethasone maintenance therapy (Kd) or observation. The primary clinical endpoint was time to progression, which was extended by 8 months by Kd. The aim of this paper is to present the all HRQL endpoints of the CARFI trial including the HRQL effect of Kd maintenance therapy relative to observation. The primary HRQL endpoint was assessed by EORTC QLQ-C30 Summary score (QLQ-C30-sum) at 8 months follow-up. A key secondary HRQL endpoint was quality-adjusted progression-free-survival (QAPFS). Methods HRQL was assessed with EORTC QLQ-C30, EORTC QLQ-MY20 and FACT/GOG-Ntx at randomization and every second month during follow-up. HRQL data were analyzed with linear mixed effect models until 8 months follow-up. QAPFS per individual was calculated by multiplying progression-free survival (PFS) by two quality-adjustment metrics, the QLQ-C30-sum and EORTC Quality of Life Utility Measure-Core 10 dimensions (QLU-C10D). The QAPFS per treatment group was estimated with the Kaplan-Meier method. P < 0.05 was used for statistical significance, and a between-group minimal important difference of 10 points was interpreted as clinically relevant for the QLQ-C30-sum. Results 168 patients were randomized. HRQL questionnaire compliance was 93%. For the QLQ-C30-sum, the difference of 4.62 points (95% confidence interval (CI) -8.9: -0.4, p = 0.032) was not clinically relevant. PFS was 19.3 months for the Kd maintenance group and 16.8 months for the observation group; difference = 2.5 months (95% CI 0.5; 4.5). QAPFS based on the QLQ-C30-sum for the Kd maintenance group was 18.0 months (95% CI 16.4; 19.6) and for the observation group 15.0 months (95% CI 13.5; 16.5); difference = 3.0 months (95% CI 0.8–5.3). QAPFS based on the QLU-C10D for the Kd maintenance group was 17.5 months (95% CI 15.9; 19.2) and 14.0 months (95% CI 12.4; 15.5) for the observation group; difference = 3.5 months (95% CI 1.1–5.9). Conclusions Kd maintenance therapy after salvage ASCT did not adversely affect overall HRQL, but adjustment for HRQL reduced the PFS compared to unadjusted PFS. PFS of maintenance therapy should be quality-adjusted to balance the benefits and HRQL impact.

Conceptualising 'Benefits Beyond Health' in the Context of the Quality-Adjusted Life-Year: A Critical Interpretive Synthesis

Progression-free (PFS) and overall survival (OS) landmark analysis from

Multiple Myeloma, Myeloma, Multiple Myeloma Symptoms

The quality-adjusted life-years in the oncological patients' health-related quality of life
Frontiers Children of Mentally III Parents at Risk Evaluation (COMPARE): Design and Methods of a Randomized Controlled Multicenter Study—Part I

Effect sizes and relative validities for the scales and summary score

OUH - Group members

Quality-adjusted life year is useful, not discriminatory- STAT

Disparities in Health and Health Care: 5 Key Questions and Answers

Health-related quality of life in the ENDEAVOR study: carfilzomib-dexamethasone vs bortezomib-dexamethasone in relapsed/refractory multiple myeloma

OUH - Group members

The application of the drug user quality of life scale (DUQOL) in Australia

PDF) Expanded Prostate Cancer Index Composite for Clinical Practice: Development and Validation of a Practical Health Related Quality of Life Instrument for Use in the Routine Clinical Care of Patients With Prostate

PDF) Health-Related Quality of Life Results From the Open-Label, Randomized, Phase III ASPIRE Trial Evaluating Carfilzomib, Lenalidomide, and Dexamethasone Versus Lenalidomide and Dexamethasone in Patients With Relapsed Multiple Myeloma