Color change is only device modification. Is a new 510k required? - Medical Device Academy
$ 12.00
In stock
4.7
(77)

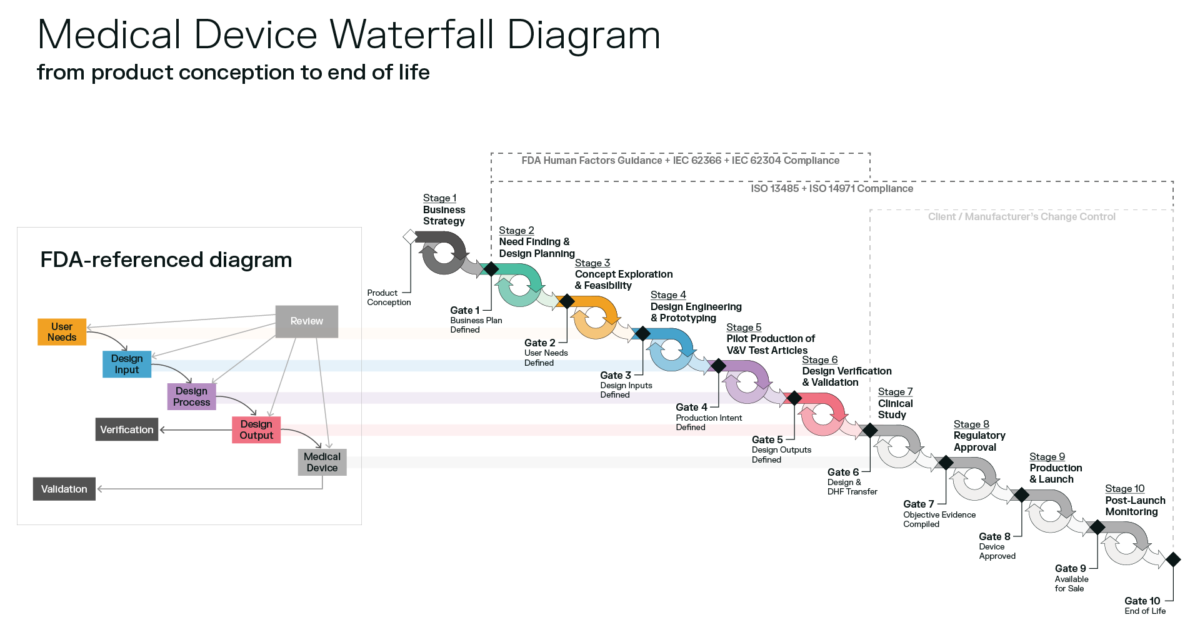
This article explains the process for determining if a color change and other material changes require a new 510k prior to implementing the change.
cms./assets/images/_1200x630_crop_center
FDA

Substantial Equivalence - an overview
FDA

FDA Guidance on 510(k) for Changes to Existing Devices

Your Medical Devices Are Getting Smarter. Can the FDA Keep Them Safe? - WSJ
FDA

What Should be Included in a 510k

Substantial Equivalence - an overview

Does My Modified Medical Device Require a New 510(k)?

/sites/default/files/field/image/