An ideal gas initially P_i ,V_i , and T_i is taken through a cycle

Click here:point_up_2:to get an answer to your question :writing_hand:an ideal gas initially at pi vi and ti is taken through a cycle
Click here👆to get an answer to your question ✍️ An ideal gas initially P-i -V-i - and T-i is taken through a cycle as shown in Figure- -a- Find the net work done on the gas per cycle 1-00 mol of gas initially 0-0C- -b- What is the net energy added by heat to the gas per cycle
1st law

Revision Thermodynamics 2021-SV, PDF, Gases

Heat and the First Law of Thermodynamics

Solved An ideal gas initially at Pi, V;, and T; is taken

An ideal gas initially at pressure P0, volume V0, and temperature T0 is taken through the cycle described in Figure P12.54, with n = 4 and m = 3. Figure P12.54 (a)

An ideal gas initially P_i ,V_i , and T_i is taken through a cycle as shown in Figure. (a) Find the net work done on the gas per cycle 1.00 mol of

11/14/2013PHY 113 C Fall Lecture 221 PHY 113 C General Physics I 11 AM – 12:15 PM MWF Olin 101 Plan for Lecture 22: Chapter 21: Ideal gas equations. - ppt download

Thermodynamics: An Engineering Approach - 5th Edition - Part II by 黑傑克 - Issuu

One mole of a gas in the state A(P1, V1, T1) is subjected to adiabatic

1st law

Thermodynamics problems
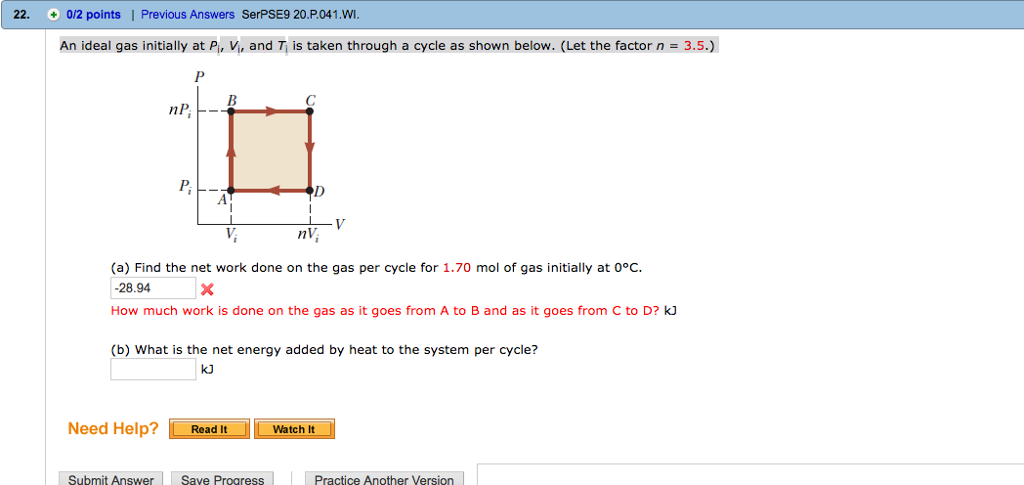
Solved An ideal gas initially at Pi, Vi, and Ti is taken

Heat and the First Law of Thermodynamics